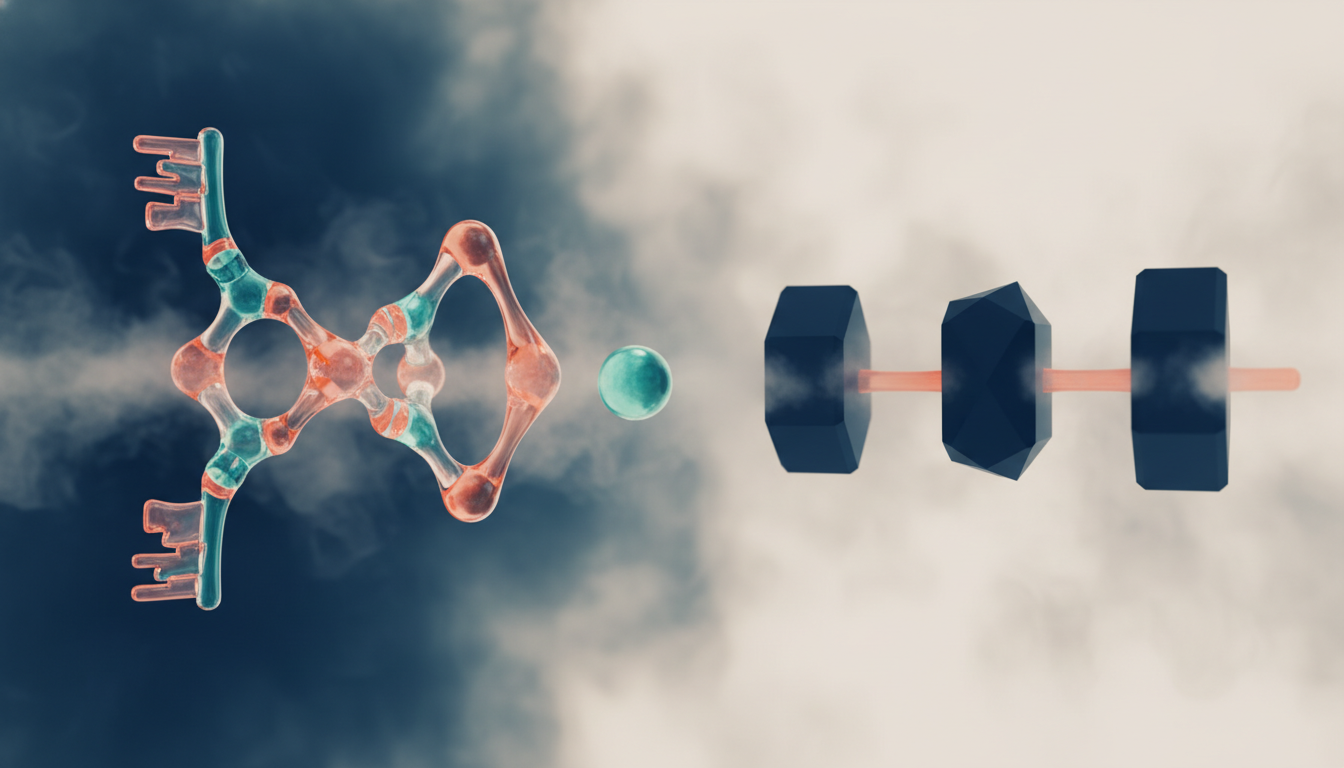
Peptides vs small-molecule drugs: keys vs hammers
Two very different strategies for influencing biology — and the distinction matters more than most people realize.
TL;DR
- Small-molecule drugs are chemically simple, orally bioavailable, and often interact with multiple biological targets — sometimes intentionally, sometimes not.
- Peptides are larger, more structurally complex molecules that typically bind to one specific receptor — more like a key shaped for a single lock.
- Sermorelin, for example, binds primarily to the GHRH receptor in the pituitary; that specificity is a defining feature of peptide pharmacology.
What it is
A small-molecule drug is typically a synthetic compound with a molecular weight under 500 Daltons — small enough to cross cell membranes, survive digestion, and be taken orally. Aspirin, statins, and most psychiatric medications fall into this category. Their small size lets them access parts of the body that larger molecules cannot, but it also means they can interact with a broad range of biological targets.
Peptides are larger (typically 500 to 5,000 Daltons for the compounds used in clinical contexts) and structurally more complex. That size is what makes most peptides injectable rather than oral — the digestive system breaks peptide bonds before they can reach the bloodstream intact.
How it works
Think of a receptor as a lock and a signaling molecule as a key. Peptides are shaped to fit one specific lock. Small molecules, because of their simple structure, sometimes work more like a hammer — they can bang on many locks at once, which is why small-molecule drugs often carry longer lists of off-target effects.
Receptor specificity research published in Nature Reviews Drug Discovery has highlighted that the structural complexity of peptides gives them an inherent targeting advantage for receptor-level interactions (Fosgerau & Hoffmann, Nature Reviews Drug Discovery, 2015). That same study notes that over 60 peptide drugs had received regulatory approval globally by 2015, with more than 140 in active clinical trials at the time.
Sermorelin illustrates this well. It is a synthetic version of the first 29 amino acids of growth-hormone-releasing hormone (GHRH). Its primary binding target is the GHRH receptor on somatotroph cells in the anterior pituitary — a defined, well-characterized interaction studied in peer-reviewed literature for decades.
Who asks about it
This question comes up most often from people who have done some research and want to understand why peptides are often described as more targeted than conventional pharmaceuticals — and whether that claim holds up. It also comes up from anyone navigating the difference between, say, an oral hormone precursor supplement (small molecule) and a peptide prescribed through a telehealth clinician.
What the research says
The Fosgerau & Hoffmann review remains one of the most-cited overviews of peptide therapeutics in the published literature. The authors identify receptor specificity as a primary rationale for peptide drug development, noting that the “exquisite selectivity” of peptides for their target receptors often translates to a more predictable interaction profile compared to promiscuous small molecules. They also acknowledge the trade-offs: shorter half-lives, injection routes, and manufacturing complexity.
A subsequent review in Pharmacological Reviews (Muttenthaler et al., 2021) expanded this picture, documenting over 80 approved peptide therapeutics and emphasizing that advances in half-life extension (such as PEGylation and fatty-acid conjugation) are narrowing the practical gap between peptides and traditional drugs (Muttenthaler et al., Pharmacological Reviews, 2021).
What to know before considering it
Receptor specificity is a property of the molecule — it does not guarantee a particular clinical outcome for any individual. Peptides require a prescription, clinician evaluation, and appropriate lab work before use. The pharmacology is a reason researchers find peptides interesting; it is not a substitute for individualized medical assessment.
The Halftime POV
The keys-vs-hammers frame isn’t just a clever metaphor — it reflects a real structural difference in how these molecules interact with biology. That specificity is part of why peptide research has attracted serious scientific attention over the past three decades. Understanding it helps you ask better questions of your clinician rather than walking in with a compound name and a wish list.
Related reading:
FAQ
Q: How do peptides differ from small-molecule drugs? A: Peptides are larger molecules (typically 500–5,000 Daltons) that bind to specific receptors with high precision — like a key in a single lock. Small-molecule drugs are smaller (under 500 Daltons), often orally bioavailable, and frequently interact with multiple biological targets simultaneously, which can increase off-target effects.
Q: Why are most peptides injected instead of taken orally? A: Peptide bonds are broken down by digestive enzymes before reaching the bloodstream, making oral bioavailability negligible for most therapeutic peptides. Subcutaneous injection bypasses the digestive system entirely, delivering the compound directly to systemic circulation via the fatty tissue layer below the skin.
Q: Is receptor specificity a guarantee of safety? A: No. Receptor specificity means a peptide tends to have a narrower, more predictable interaction profile compared to promiscuous small molecules — but it does not guarantee a particular safety outcome for any individual. All peptide use requires clinician evaluation, appropriate labs, and ongoing monitoring.
Disclaimer
This article is educational and is not medical advice. Compounded medications are not FDA-approved. Clinical outcomes depend on individual factors and require physician evaluation. Results vary. Halftime Health is launching soon — join the waitlist to get updates.
Get updates
Halftime Health is launching soon. We’ll share what we learn along the way — the research, the regulations, the real-world trade-offs. Join the waitlist and we’ll email you when we’re live.
Sources
- Fosgerau K, Hoffmann T. Peptide therapeutics: current status and future directions. Nature Reviews Drug Discovery, 2015.
- Muttenthaler M, et al. Trends in peptide drug discovery. Pharmacological Reviews, 2021.